Battery life span and degradation
One of the most common questions from customers considering home battery storage is "How long will it last?"
Batteries in phones and laptops noticeably degrade within a few years: the smartphone that once lasted all day dies by mid-afternoon. So why should a home battery be any different?
The answer lies in chemistry, design, and management. Home storage batteries use different chemistry to your phone, operate under much gentler conditions, and include management systems to maximise their lifespan. Understanding the science behind battery degradation explains why a well-designed home battery system should comfortably outlast its warranty and continue delivering value for many years beyond.
How lithium-ion batteries work
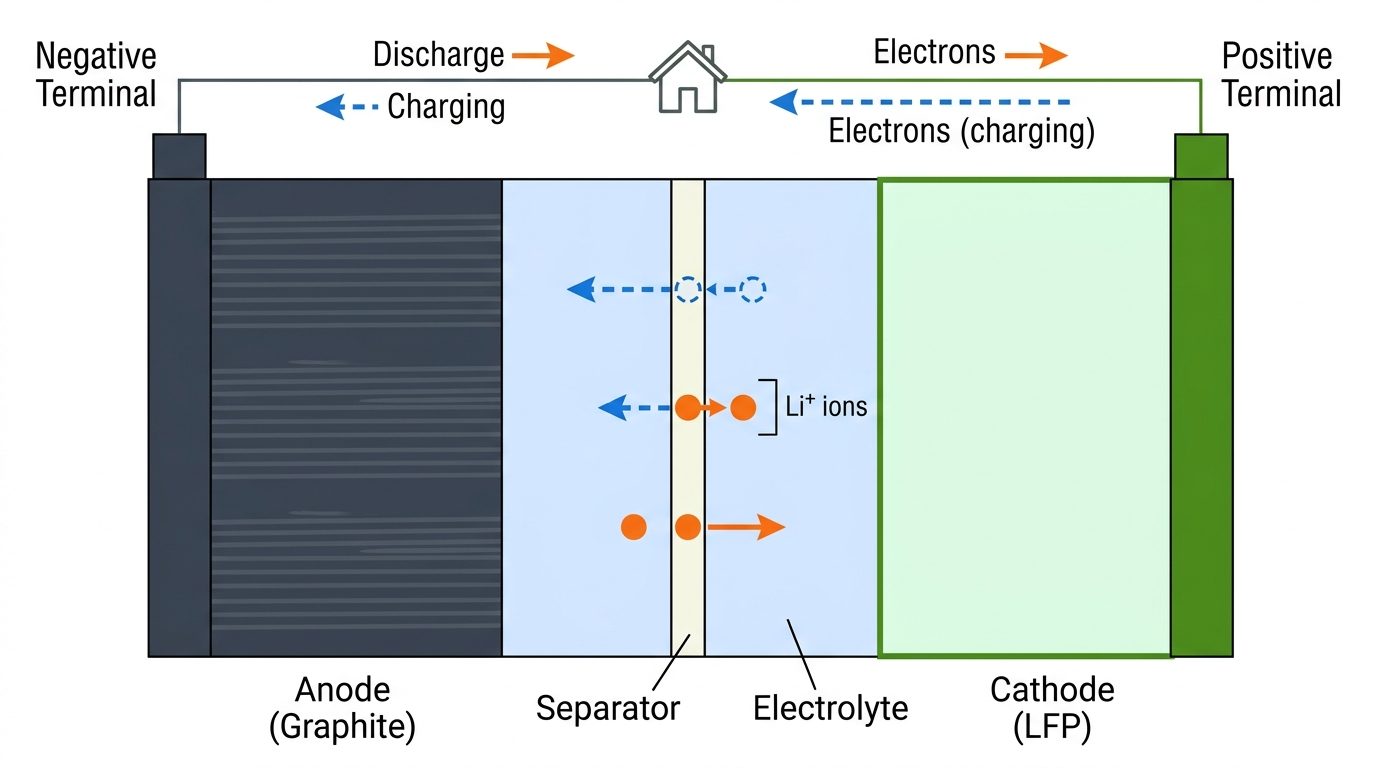
A lithium-ion cell has four main components: a negative electrode (anode), a positive electrode (cathode), an electrolyte, and a separator. The anode is typically graphite; the cathode material varies by chemistry: this is what distinguishes “LFP” from “NMC” and others.
When you charge a battery, lithium ions leave the cathode, travel through the electrolyte, and slot into the layered structure of the graphite anode. When you discharge, the ions make the reverse journey. This process is called intercalation, ions insert themselves into the electrode structure without fundamentally changing it, like sliding books onto a shelf. The electrolyte conducts lithium ions but not electrons, which forces electrons through the external circuit to power your home.
This system can repeat thousands of times, but not indefinitely. Each cycle causes small amounts of wear, and understanding those mechanisms is key to understanding battery longevity.
Schematic of a typical cell and how electrons and lithium ions behave during charging and discharging.
The main lithium-ion chemistries
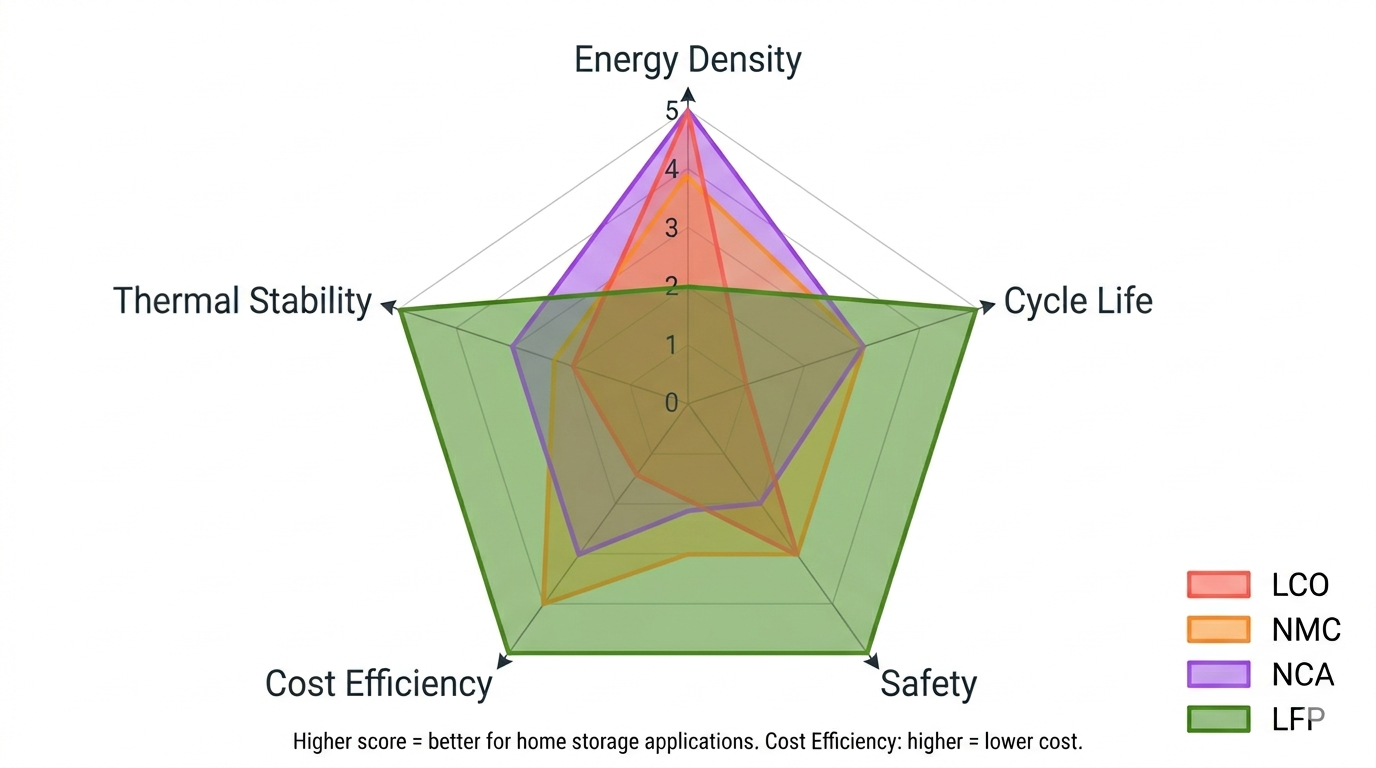
The cathode material determines many of a battery's key characteristics: energy density, cycle life, safety, and cost. Here's how the major chemistries compare.
LCO (Lithium Cobalt Oxide)
The chemistry in smartphones and laptops. High energy density in a compact package, but short cycle life (500 - 1,000 cycles), limited thermal stability, and high cobalt content. Not used in home storage.
NMC (Lithium Nickel Manganese Cobalt Oxide)
Common in electric vehicles and earlier home batteries. A reasonable balance of energy density and cycle life (1,500 - 3,000 cycles), but requires careful thermal management and contains cobalt.
NCA (Lithium Nickel Cobalt Aluminium Oxide)
Used in some premium EVs. Very high energy density with similar cycle life to NMC. Also requires active thermal management and contains cobalt.
LFP (Lithium Iron Phosphate)
Lower energy density than the chemistries above, which means a battery of the same capacity will be larger and heavier. But for home storage, where weight and volume are not important constraints, this trade-off is overwhelmingly worthwhile:
Exceptional cycle life: 3,000 - 10,000+ cycles.
Excellent thermal stability: The iron-phosphate bond is extremely strong, making thermal runaway virtually impossible under normal conditions.
No cobalt: Lower cost and fewer supply chain concerns.
Flat discharge curve: Stable voltage across most of the cycle.
Slower calendar ageing: Lower reactivity means less degradation even when idle.
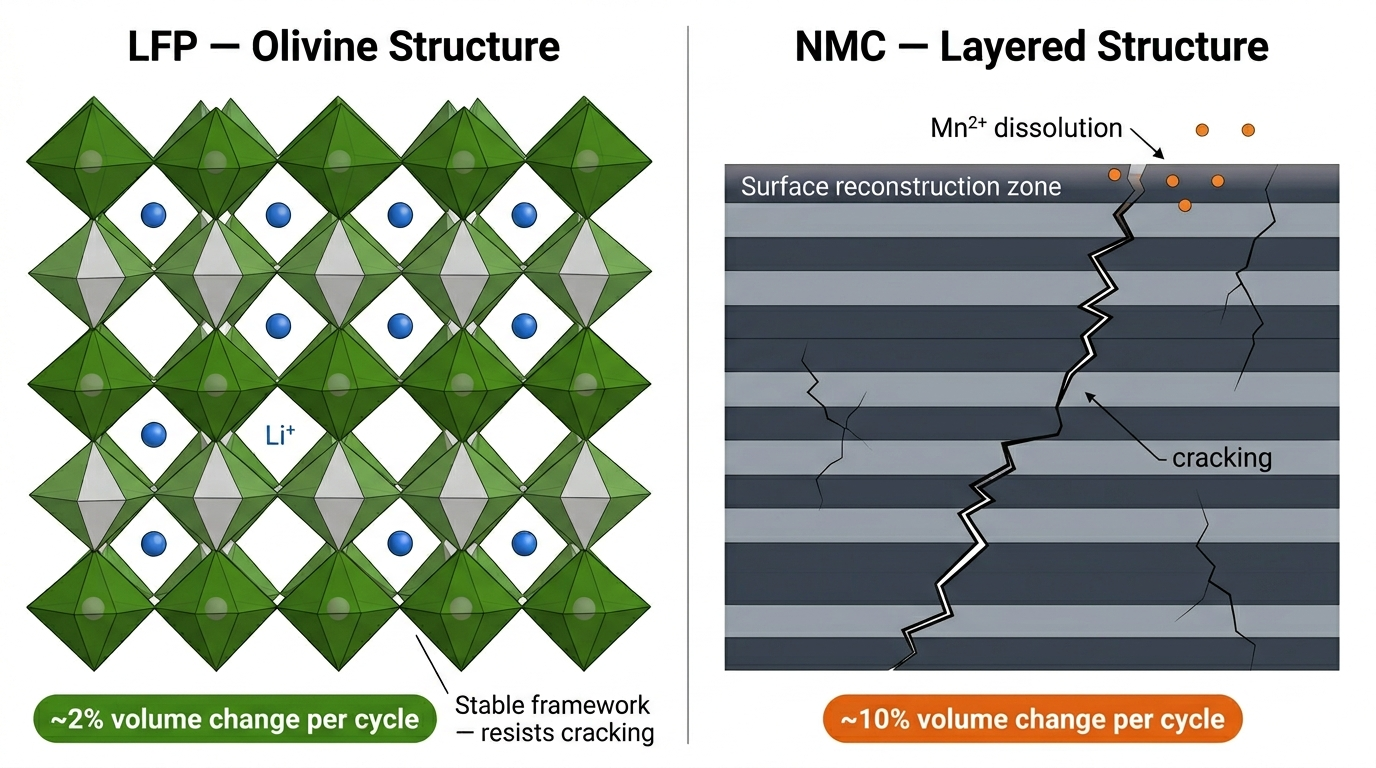
The crystal structure of LFP experiences only about 2% volume change during cycling, compared to 10% or more for layered oxide cathodes like NMC. This structural stability is a primary reason for LFP's superior longevity.
How the four main battery cathode chemistries compare on important parameters.
What causes battery degradation?
Battery degradation manifests as capacity fade (the battery holds less energy) and power fade (it can't charge or discharge as quickly). Both stem from the mechanisms below.
SEI layer formation and growth
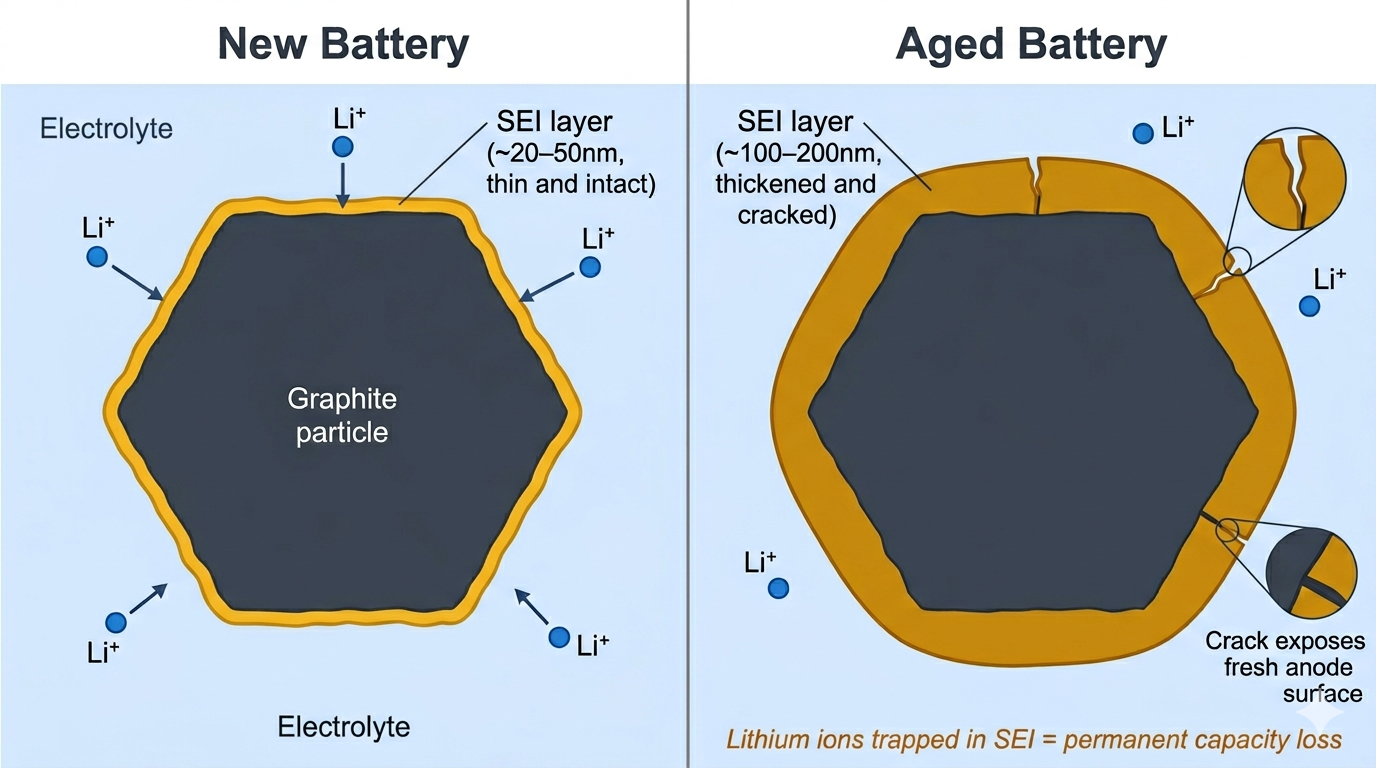
When a lithium-ion battery is first charged, the organic electrolyte reacts with the graphite anode surface and forms a thin solid layer called the Solid Electrolyte Interphase (SEI). Once formed, this layer is essential: it protects the anode from further decomposition while still allowing lithium ions to pass through.
The problem is that the SEI continues to grow slowly throughout the battery's life, consuming lithium ions and electrolyte. Each lithium ion incorporated into the SEI can no longer participate in cycling. This is loss of lithium inventory, a primary cause of capacity fade. The SEI also isn't mechanically robust; it can crack during the volume changes that occur with each charge and discharge cycle, exposing fresh anode surface and triggering more SEI formation.
SEI growth accelerates at higher temperatures and higher states of charge. LFP's lower operating voltage (3.2V nominal vs 3.6–3.7V for NMC) creates less aggressive conditions for this process, and its minimal volume change reduces mechanical cracking of the SEI layer.
The SEI layer grows and impacts battery performance over time.
Lithium plating
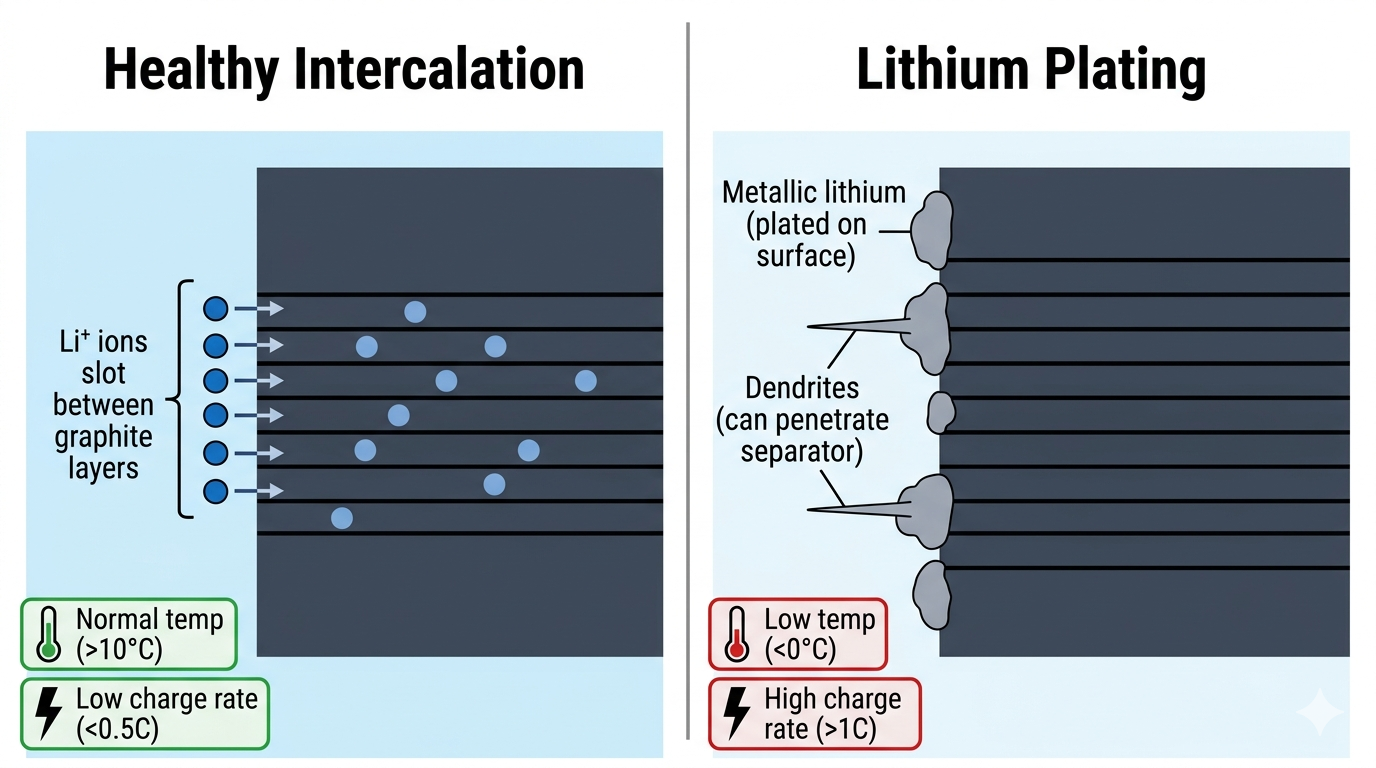
Under certain conditions, lithium ions arriving at the anode during charging cannot intercalate into the graphite quickly enough, and instead deposit as metallic lithium on the surface. This lithium plating is one of the most damaging degradation mechanisms.
Plating occurs primarily when:
Charging too fast: High current pushes ions faster than the anode can accommodate them.
Charging at low temperatures: Cold conditions slow intercalation kinetics significantly, the risk increases below ~10°C and becomes severe below 0°C.
Charging at high state of charge: Fewer available intercalation sites.
Plated lithium is lost from the active cycle (a direct capacity hit), immediately forms additional SEI, and in severe cases can form dendrites: needle-like metallic structures that can penetrate the separator. Plating is almost entirely preventable through proper management: moderate charge rates, avoiding cold-temperature charging, and managing state of charge within sensible bounds.
Rapid charging and low temperatures can lead to damaging lithium plating on the anode surface.
Cathode degradation
The cathode experiences distinct degradation pathways, particularly in layered oxide chemistries (NMC, NCA):
Particle cracking: Repeated volume changes cause mechanical fracturing, creating new electrolyte-exposed surfaces and isolating particles from the electrode.
Transition metal dissolution: Manganese can dissolve into the electrolyte and migrate to the anode, accelerating SEI growth.
Phase transitions: Charging above ~80% State of Charge (SoC) in NMC/NCA pushes the material into a less stable structural phase. That is a key reason EV manufacturers recommend against regular 100% charging.
LFP's crystal structure avoids all of these mechanisms. Its ~2% volume change eliminates the particle cracking that plagues layered oxides. Iron dissolution is minimal and the structure remains stable across the entire operating range without phase transitions.
The crystal structure of LFP suffers from less volumetric change than layered Nickel oxides.
Calendar ageing
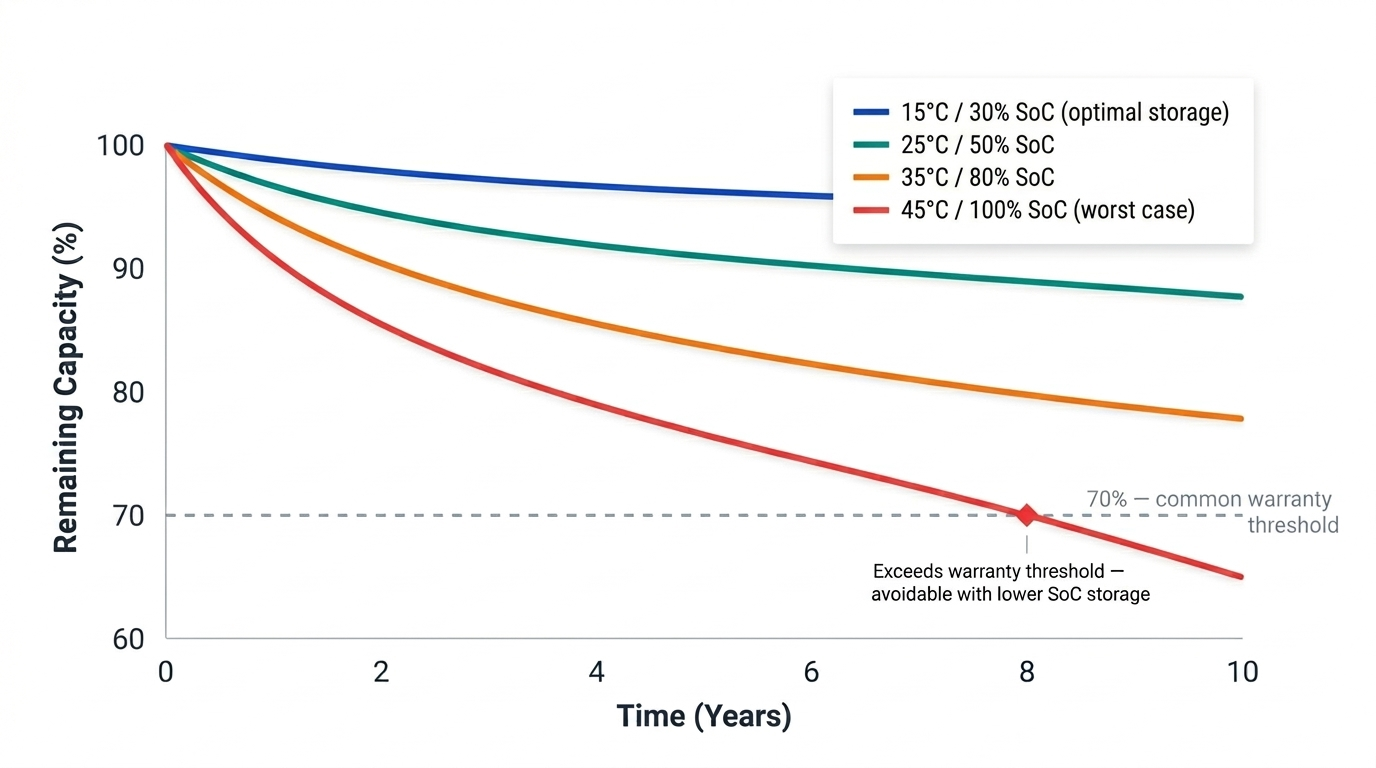
Batteries degrade even when idle. This calendar ageing is driven primarily by continued SEI growth, which proceeds slowly regardless of cycling. The rate depends strongly on temperature and state of charge (SoC): a battery stored hot and fully charged ages much faster than one stored cool at moderate SoC.
Calendar ageing roughly follows a square-root-of-time relationship: capacity loss decelerates over time under constant conditions. For home storage, this is relevant during extended periods away from home and another reason intelligent charge management can usefully improve lifetime during prolonged idle periods.
How temperature and state of charge (SoC) can impact calendar ageing.
How operating conditions affect longevity
Heat
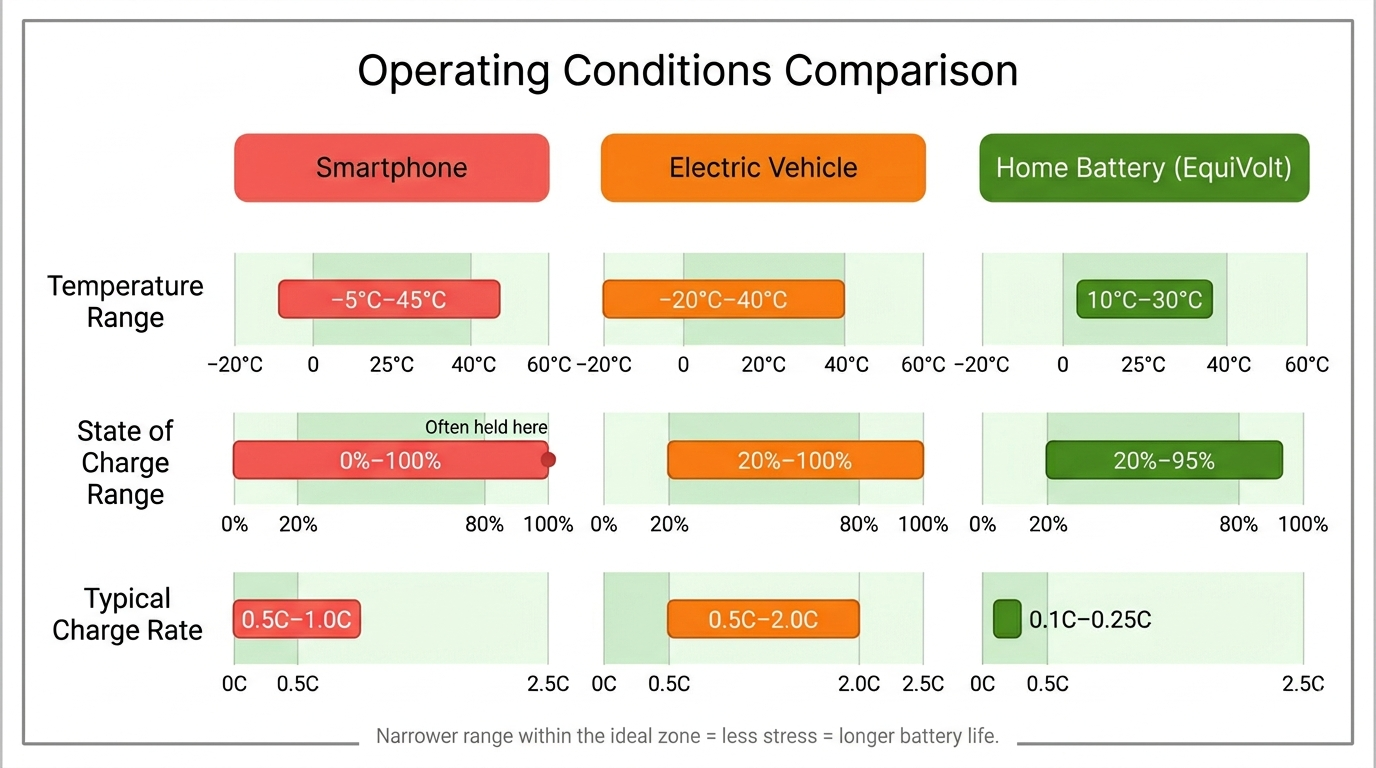
Temperature is the single most important factor. As a rough rule, every 10°C increase roughly doubles the rate of most degradation mechanisms. Phone batteries experience daily temperature swings from pocket warmth to outdoor cold, plus charging heat. UK home batteries, installed indoors or in a sheltered environment, typically see stable temperatures of 10 - 30°C, near-ideal conditions.
Depth of discharge
Deeper cycles cause more mechanical stress on electrodes and push voltages closer to the extremes where degradation accelerates. Home energy arbitrage naturally tends toward partial cycles. A 15kWh battery providing 10kWh of daily shifting operates at roughly 67% depth of discharge, which is comfortably within the range that maximises longevity.
Charge rate
Higher charge rates generate more heat and increase lithium plating risk. The charge rate is often referred to in terms of “C”: the charge rate that would fill the battery in 1-hour. A rate of 0.5C would charge a battery in 2-hours, 2C would charge it to full in 30 minutes. Home storage typically charges slowly: EquiVolt systems often charge over a 5-hour off-peak window, typically at rates <0.2C, which is exceptionally gentle compared to the 1 - 2C rates of EV rapid charging.
Time at extreme state of charge
High SoC drives more parasitic reactions. Very low SoC raises internal resistance and risks copper dissolution from the anode current collector. A well-managed system spends most of its time comfortably between these extremes.
How different operating conditions for battery applications compare.
How modern systems maximise lifespan
Battery Management System (BMS)
Every cell in the pack is monitored by a Battery Management System that tracks voltage, current, and temperature, ensuring operation stays within safe limits. Key functions include overcharge and overdischarge protection, overcurrent limiting, and temperature monitoring.
The BMS also handles cell balancing, which ensures all cells in the pack age evenly. Balancing occurs automatically when the system reaches a high state of charge (near 100%), where the BMS can redistribute charge between cells and compensate for any small differences that have developed over time. Left unmanaged, these small imbalances compound over time, with weaker cells limiting the whole pack's performance.
Internal heating
EquiVolt batteries include internal heating elements that activate automatically when cell temperature drops below the threshold for safe charging. In UK winters, particularly in garages or outbuildings where temperatures can fall near or below freezing overnight, this protection is important. The heater brings cells to a safe operating temperature before charging begins, which prevents the lithium plating that cold charging would otherwise risk. This is a hardware feature that operates automatically, requiring no configuration.
EquiVolt+: Intelligent charge management
Beyond the hardware-level protections, the EquiVolt+ subscription adds a software layer that actively optimises charge patterns for both financial return and battery longevity.
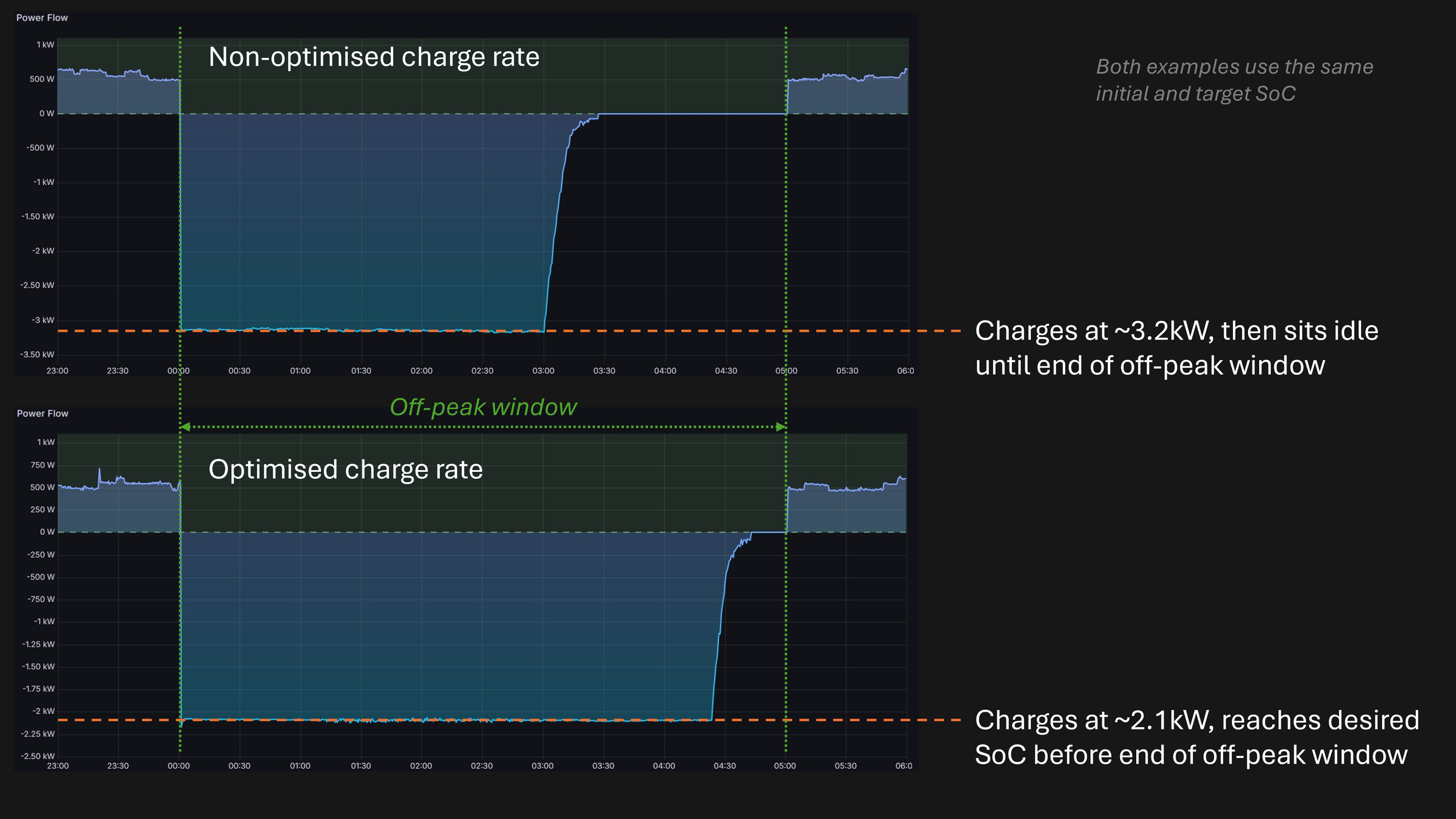
Minimising charge rate while maximising arbitrage. The most straightforward approach to overnight charging would be to charge as fast as possible at the start of the off-peak window. EquiVolt+ takes a different approach: it intelligently spreads charging across the available off-peak period, using the slowest rate consistent with reaching the target state of charge before the window closes. This minimises heat generation and reduces lithium plating risk, meaningfully extending cycle life but without sacrificing any arbitrage value, since the battery still reaches the same charge level by morning.
Periodic cell balancing. Because BMS balancing only occurs near 100% SoC, a system that never reaches full charge (for example, one conservatively capped at 90%) may go extended periods without triggering a balance cycle. EquiVolt+ periodically schedules a full charge to deliberately activate the BMS balancing routine. This keeps cell-to-cell variation in check without requiring the battery to sit at 100% routinely.
State of charge optimisation. The system avoids holding the battery at 100% for extended periods unnecessarily, adjusts cycle depth based on actual usage patterns, and can reduce SoC during prolonged idle periods to limit calendar ageing.
Together, these management strategies can meaningfully extend effective battery life compared to simple charge-discharge strategies. This translates into more value over the system's lifetime, not just lower bills in the short term.
Real data showing non-optimised and optimised charging profiles from an EquiVolt pilot programme system. Data shared with permission.
Longevity: what to expect
Warranty terms
EquiVolt batteries carry a 10-year or 8,000-cycle warranty, guaranteeing at least 70% retained capacity, whichever limit is reached first.
To contextualise those numbers: 8,000 cycles at one cycle per day equates to approximately 22 years. In practice, home batteries often perform partial cycles, so cycle count accumulates more slowly. The 10-year calendar limit accounts for ageing regardless of cycle count. A 15kWh system at 70% capacity still provides 10.5kWh of usable storage, which is sufficient for most household arbitrage applications.
Beyond the warranty
Degradation is gradual, not a cliff edge. A well-maintained LFP system reaching 70% capacity at year 10 might retain 60% at year 15 and perhaps 50% at year 20, which still delivers meaningful daily arbitrage value. Field data from large-scale LFP deployments increasingly supports cycle life figures at the high end of laboratory predictions: 6,000 - 10,000+ cycles to 80% capacity under real-world conditions.
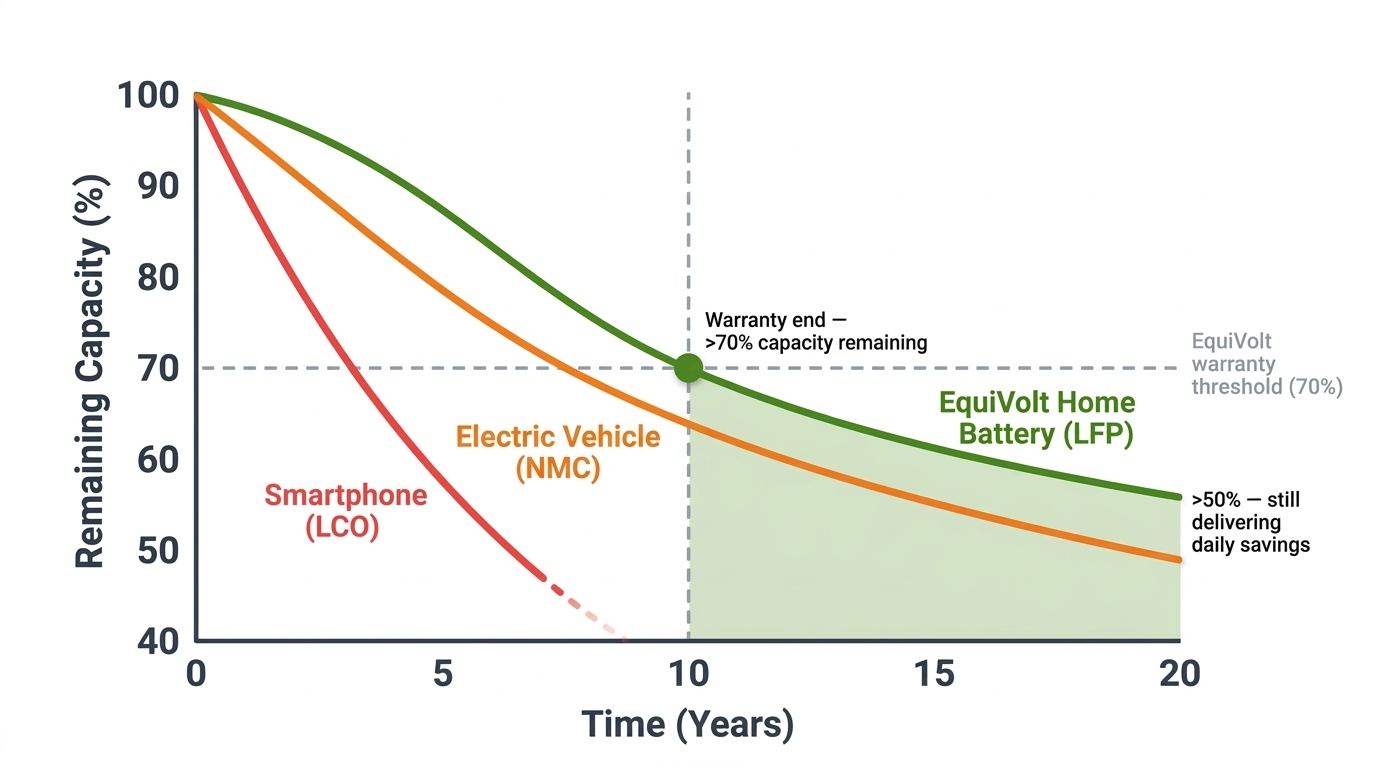
Comparison with other applications
The same underlying technology shows dramatically different longevity depending on chemistry and operating conditions. Home storage represents the ideal combination: the most durable chemistry operating under the gentlest conditions.
How the longevity of typical battery applications compare.
Conclusion
LFP chemistry offers real advantages for longevity: a stable crystal structure that resists cracking, minimal volume change during cycling, lower operating voltages, and no problematic phase transitions. Those chemical advantages are reinforced by hardware protections (BMS, internal heating) and, with EquiVolt+, by intelligent charge management that minimises stress on the battery while preserving full arbitrage value.
References
U.S. Department of Energy: How lithium ion batteries work
Battery Design: Lithium ion chemistry
Journal of The Electrochemical Society: Calendar Aging of Lithium-Ion Batteries
NREL: Life Prediction Model for Grid-Connected Li-ion Battery Energy Storage System